Translatomics for translational control insights
Recent Publications Harnessing the Power of Translatomics
Every week we provide a digest of a small number of recent interesting papers in the field of translatomics.
In this week’s Sunday papers,
- Hwang et al. reveal YAP/TAZ maintain translation under starvation via Ribo-seq and polysome profiling.
- Musalgaonkar et al. use selective Ribo-seq to track Reh1 dynamics during early elongation.
- Liu et al. demonstrate YTHDF2 promotes translation of m6A RNAs, confirmed by polysome profiling.
Together, these studies highlight how ribosome occupancy and translation regulation control protein output in health and disease.
YAP promotes global mRNA translation to fuel oncogenic growth despite starvation
Experimental & Molecular Medicine, 2024.
Daehee Hwang, Seonguk Baek, Jeeyoon Chang, Taejun Seol, Bomin Ku, Hongseok Ha, Hyeonji Lee, Suhyeon Cho, Tae-Young Roh, Yoon Ki Kim, Dae-Sik Lim
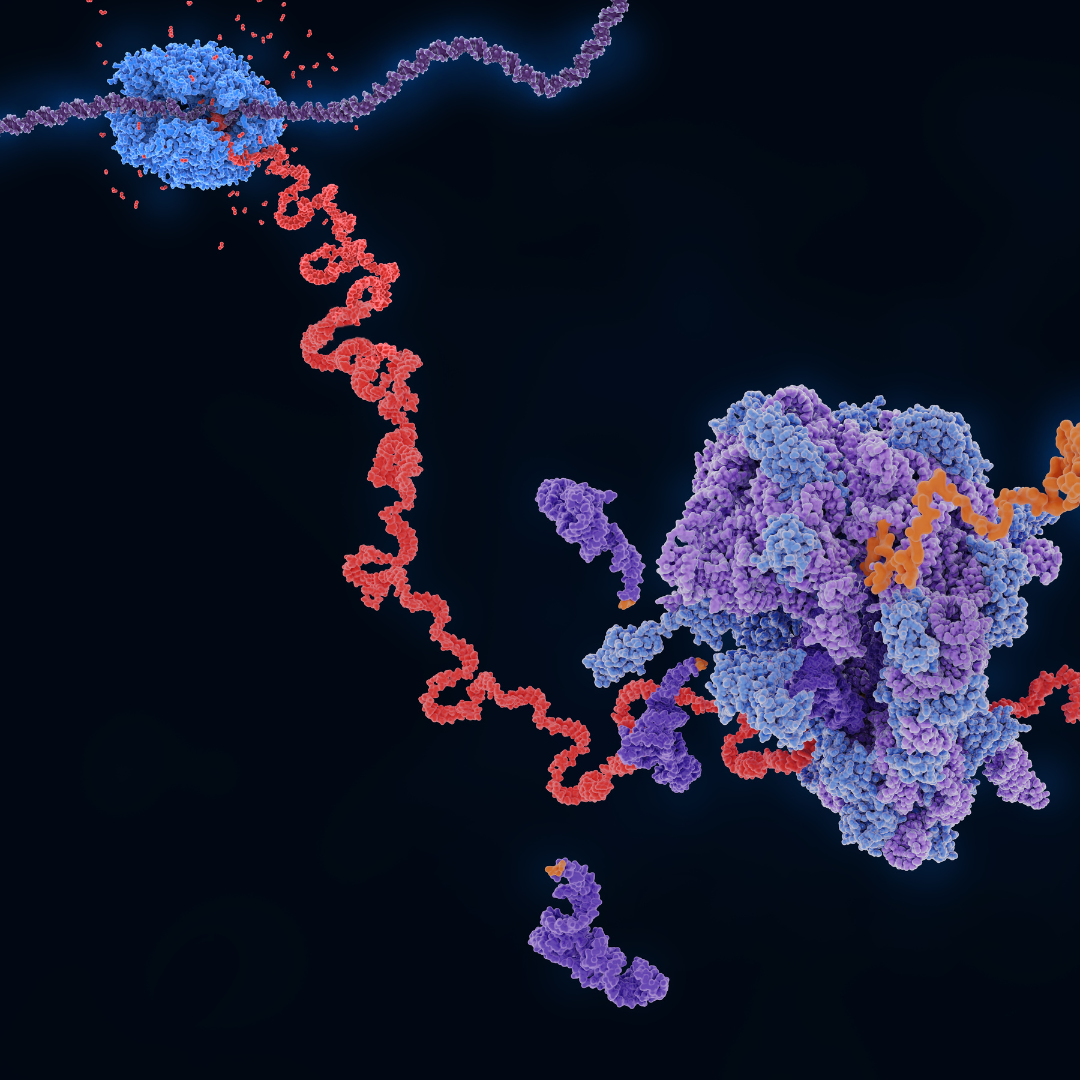
This study investigates how the oncogenic coactivators YAP/TAZ maintain global protein synthesis under serum starvation, a condition that normally suppresses translation. The authors employed ribosome profiling (Ribo-seq) to measure genome-wide ribosome occupancy and polysome profiling to assess the distribution of mRNAs across translating ribosomes, confirming that YAP/TAZ preserve heavy polysomes.
YAP/TAZ activation restored translation of 5′TOP mRNAs, including ribosomal proteins and translation factors, via mTORC1 signaling. YAP-induced translation then increased proliferation under starvation and could be reversed by restoring DDIT4 (an mTORC1 inhibitor) or by directly inhibiting mTORC1.
The authors demonstrated that translational regulation is central to oncogenic growth under stress. The combination of Ribo-seq and polysome profiling provides a detailed mechanistic insight into how cancer cells sustain protein synthesis, and points to mTORC1-dependent translation as a potential therapeutic vulnerability in YAP/TAZ-driven tumours.
Learn more about EIRNABio’s ribosome profiling and polysome profiling services here.
The assembly factor Reh1 is released from the ribosome during its initial round of translation
Nature Communications, 2025.
Sharmishtha Musalgaonkar, James N. Yelland, Ruta Chitale, Shilpa Rao, Hakan Ozadam, David W. Taylor, Can Cenik, Arlen W. Johnson

This study explores ribosome maturation and early translation using selective ribosome profiling, a variant of Ribo‑Seq. The authors investigated Reh1, an assembly factor thought to dissociate prior to translation and tracked its association with ribosomes during the first rounds of translation.
By isolating Reh1-bound ribosomes and sequencing ribosome-protected fragments (RPFs), they found that Reh1 remains bound during early elongation, with RPFs enriched near start codons. Cryo-EM confirmed Reh1 occupies the ribosomal exit tunnel, and its release is coupled to nascent chain elongation. Truncation experiments on the Reh1 C-terminus support a model in which the elongating nascent chain displaces Reh1 from the exit tunnel as translation progresses.
This paper demonstrates that selective Ribo‑Seq can reveal transient ribosome states, linking ribosome maturation to translation initiation and underscores the utility of ribosome profiling for mapping early translational events.
Learn more about EIRNABio’s ribosome profiling services here.
The RNA-stability-independent role of the RNA m6A reader YTHDF2 in promoting protein translation to confer tumor chemotherapy resistance
Molecular Cell, 2025.
Tao Liu, Dan Yang, Qinglv Wei, Yuan Wang, Lei Tian, Xiaoyi Liu, Yu Yang, Qingya Luo, Jie Xu, Yujiao Liu, Chenyue Yang, Xinzhao Zuo, Fatao Luo, Xin Luo, Hongyan Zhao, Li Li, Jing Xu, Jia Yu, Shoubao Ma, Jianhua Yu, Ping Yi

The authors reveal that the m6A reader YTHDF2, classically known for mRNA decay, instead enhances translation of specific m⁶A-modified mRNAs, promoting tumor chemotherapy resistance in ovarian cancer.
Using polysome profiling, they demonstrated increased ribosome occupancy on YTHDF2 target mRNAs in cancer cells. YTHDF2 interacts with eIF3F and the RNA helicase DDX1 to facilitate translation initiation, independent of mRNA stability. Functional assays, including knockdown and pharmacological inhibition, reduced translation of targets and restored chemosensitivity, confirming the biological impact.
This study highlights a critical translational control mechanism in cancer, showing how ribosome engagement can be regulated independently of mRNA abundance. It also provides a clear example of how polysome profiling can reveal regulatory mechanisms relevant to oncology and drug resistance.
Learn more about EIRNABio’s polysome profiling services here.




