Translatomics for immunology, tRNA, and NAT10
Recent Publications Harnessing the Power of Translatomics
Every week we provide a digest of a small number of recent interesting papers in the field of translatomics.
In this week’s Sunday papers,
- Dopler et al. demonstrate the role of P-stalk ribosomes in mediating cytokine-based immune responses.
- Liu et al. uncover the importance of the ac4C12 tRNA modification in serine and leucine codon translation.
- Sun et al. reveal the mechanism of NAT10-mediation mRNA modification in RNA stability during viral infection.
P-stalk ribosomes act as master regulators of cytokine-mediated processes
Cell, 2024
Dopler, A., Alkan, F., Malka, Y., van der Kammen, R., Hoefakker, K., Taranto, D., Kocabay, N., Mimpen, I., Ramirez, C., Malzer, E. and Isaeva, O.I et al
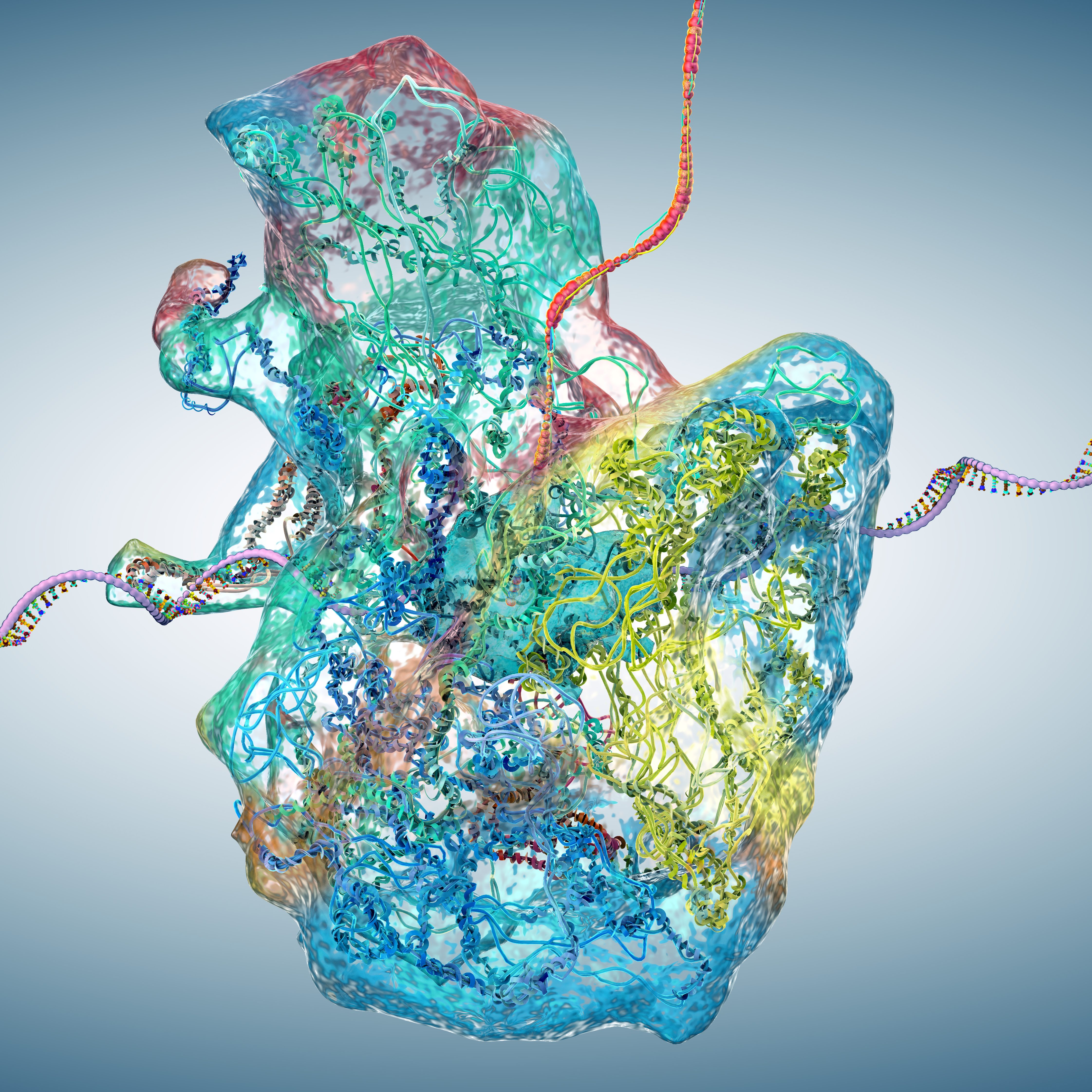
Cancer cells often evade the immune system, a process now recognized as a hallmark of cancer. While immune checkpoint blockade (ICB) therapies have shown success, many patients fail to respond or develop resistance to the therapy. Cytokines like IFNγ and TNF-α, produced by T cells, strongly influence tumour behaviour by altering gene expression, antigen presentation, and immune signaling. However, most studies focus on transcriptional changes, overlooking post-transcriptional regulation. Emerging research suggests ribosomes—the cell’s protein factories—may vary in composition and function, potentially shaping how cells respond to cytokines. This “immunoribosome” concept could reveal new mechanisms behind immune control and ICB resistance.
Researchers discovered a specialized “cytokine-responsive” ribosome, defined by increased incorporation of the P-stalk proteins P1 and P2 following exposure to immune cytokines like IFNγ and TNF-α. Using proteomics and sucrose density centrifugation, they found that actively translating ribosomes gain P1/P2 under cytokine stimulation, while inhibitory cytokines (e.g., TGF-β) reduce this incorporation. Knockdown of P1 via shRNA impaired antigen presentation (measured by HLA I flow cytometry) and T cell-mediated killing, indicating the P-stalk ribosome (PSR) regulates immune visibility. Ribosome profiling revealed that PSRs preferentially translate mRNAs for antigen processing and cytokine signaling, particularly those encoding membrane or transmembrane proteins. Phospho-proteomics showed that cytokines decrease, while TGF-β increases, phosphorylation of P-stalk proteins—modifying their ribosomal incorporation. CRISPR-edited phospho-dead mutants confirmed that blocking this phosphorylation prevents TGF-β-induced PSR inhibition. Together, these techniques revealed the PSR as a conserved, dynamically regulated ribosomal subpopulation controlling cytokine-induced immune responses.
Learn more about EIRNABio’s ribosome profiling services here.
Mammalian tRNA acetylation determines translation efficiency and tRNA quality control
Nature Communications, 2025
Liu, N., Liu, B., Ma, C.R., Cai, Z., Wang, J.T., Chai, Z.Q., Zhu, N., Shao, T., Chen, Y.L., Lin, Y. and Wang, Y. et al.
RNA molecules carry over 170 types of chemical modifications, collectively forming the epitranscriptome, which fine-tunes gene expression. One such mark, N4-acetylcytidine (ac4C), is found in tRNA, rRNA, and other RNAs, and is catalyzed by the enzyme NAT10 (or its homologs). In bacteria and archaea, ac4C enhances translation fidelity and thermal stability. In eukaryotes, it occurs mainly at position 12 of tRNA^Ser and tRNA^Leu ( ac4C12), requiring the adaptor THUMPD1. ac4C helps maintain tRNA stability, especially under heat stress. Disruption of ac4C pathways—such as NAT10 or THUMPD1 mutations—has been linked to human diseases, underscoring its role in translation and RNA quality control. Here, the authors seek to investigate the role of ac4C12 further.
Using co-immunoprecipitation (Co-IP) assays in mouse NIH/3T3 cells, researchers demonstrated that the mThumpd1 protein physically interacts with the mNat10 protein, forming an RNA/DNA-independent complex. Domain mapping of mNat10 revealed that this interaction is mediated through its N-acetyltransferase (AT) domain. Purified mThumpd1 protein was shown via electrophoretic mobility shift assays to directly bind tRNA, confirming its role as a tRNA-binding partner. CRISPR/Cas9-mediated knockout of mThumpd1 abolished ac4C modification on tRNAs but not on 18S rRNA, identifying mThumpd1 as an essential determinant of tRNA acetylation. Mass spectrometry confirmed the loss of ac4C in specific tRNASer and tRNALeu species in knockout cells, which exhibited reduced global translation and lower tRNA aminoacylation levels without affecting tRNA stability. Polysome profiling and ribosome-nascent-chain sequencing revealed impaired translation elongation, particularly at Ser and Leu codons. Dual-luciferase assays validated reduced decoding efficiency at these codons, linking mThumpd1-mediated ac4C modification to optimized tRNA function and translation fidelity.
Learn more about EIRNABio’s polysome profiling services here.
A critical role of N4-acetylation of cytidine in mRNA by NAT10 in T cell expansion and antiviral immunity
Nature Immunology, 2025
Sun, L., Li, X., Xu, F., Chen, Y., Li, X., Yang, Z., Yang, Y., Wang, K., Ren, T., Lin, Z. and Wang, H et al

To mount an effective immune response, naive T cells rapidly expand and boost protein production upon activation. While RNA levels rise modestly, protein synthesis increases dramatically, highlighting key post-transcriptional regulation. Among RNA modifications influencing this process, N4-acetylcytidine (ac4C)—installed by the NAT10 enzyme—has been shown to enhance mRNA stability and translation efficiency. Although NAT10-mediated ac4C has been linked to diseases like inflammation, aging, and cancer, its role in T cell biology remains unexplored. The authors sought to investigate this further.
NAT10 gene expression was found to be strongly upregulated during T cell activation, both in vitro and in vivo. Stimulation of naïve T cells with anti-CD3/CD28 increased NAT10 expression and global ac4C RNA acetylation, while lymphocytic choriomeningitis virus (LCMV) infection in mice revealed parallel upregulation during viral clearance. Mechanistically, NAT10 induction was driven by T cell receptor (TCR) signalling through the transcription factor c-JUN, confirmed by inhibitor studies and ChIP–qPCR. Conditional knockout (CKO) of Nat10 in T cells reduced ac4C levels, diminished splenic T cell numbers, and impaired proliferation while increasing apoptosis. RNA-seq and acRIP-seq showed that NAT10-dependent ac4C modifications stabilize and enhance translation of proliferation-related transcripts, notably Myc mRNA. Loss of NAT10 disrupted Myc expression, ribosome occupancy, and downstream metabolic programs, as verified by Ribo-seq and metabolic assays. Overexpression of NAT10 or MYC in CKO cells rescued proliferative and translational defects, establishing the NAT10–MYC axis as a key regulator of T cell growth, metabolism, and antiviral immunity.
Learn more about EIRNABio’s ribosome profiling services here.



